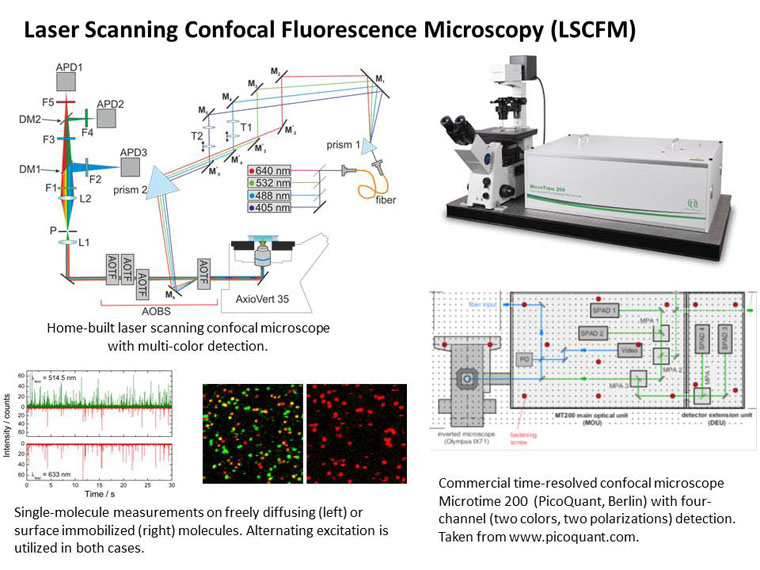
Experiments at the single-molecule level have provided novel insights into the structure and dynamics of biomolecules. Its main advantage lies in the fact that different subpopulations within a molecular ensemble can be resolved and investigated selectively. Single-pair Förster (or fluorescence) resonance energy transfer (spFRET) is a structure-sensitive spectroscopic technique that allows conformational changes to be probed in real time. Based on the coupling of optical transition dipoles of two dye molecules, referred to as the donor and the acceptor, attached to the biomolecule of interest with a mutual separation of ~20 - 100 Å, spFRET is sensitive to inter-dye distance variations in the ångström range. Thus, the technique is well suited to monitor conformational motions, for example the transition of small and medium-sized RNA and protein molecules from a completely unfolded random coil to the compact folded state.